Diet Pill Recall 2013: FDA Recalling OxyElite Pro For Links To 'Liver Illnesses And Liver Damage' [FULL LIST]
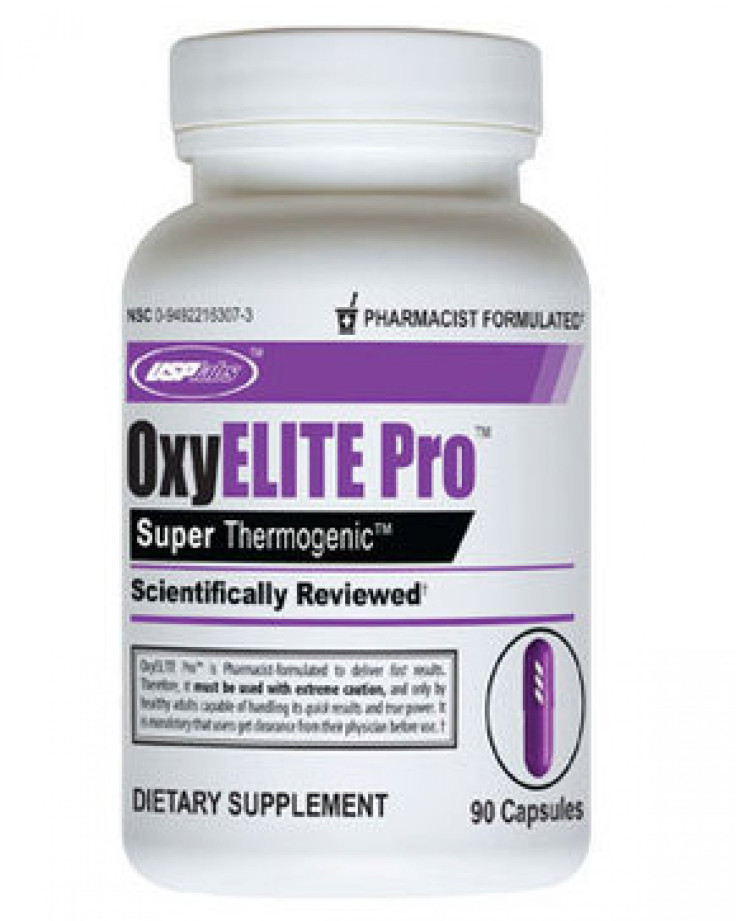
USPlabs of Dallas recalled OxyElite Pro dietary supplements from its shelves this week after the Food and Drug Administration stated the products have been linked to liver illnesses and hepatitis. The pills are used for weight loss and muscle-building.
In October, a 48-year-old woman in Hawaii who took the pills died of liver failure. Her family said she took the pills weeks before becoming violently ill. There have been dozens of other cases in which people became sick after taking the OxyElite supplement. In Hawaii, 29 cases of liver failure and acute hepatitis were reported in the last six months. It became illegal to sell OxyElite there in October. Outside Hawaii, there have been other cases of liver damage, as the products are sold all over the United States. One patient needed a liver transplant, and others are waiting for one.
In October, USPLads issued a statement concerning OxyElite and the cases of serious illness and death.
“The company stands by the safety of all of its products. The company is cooperating with FDA on reports coming out of Hawaii. The cluster of liver issues in Hawaii is a complete mystery and nothing like this has ever been associated with OxyELITE Pro in all of the years our products have been in the market. We know of no credible evidence linking OxyELITE Pro to liver issues. The ingredients have been studied for safety, are consumed in the food supply and widely used in dietary supplements. The studies and consumption history show no negative liver issues. Due to multiple products within the OxyELITE Pro brand, it may be confusing as to which products are being looked at. The specific products are the original OxyELITE Pro with DMAA, OxyELITE Pro with the “Purple Top” and OxyELITE Pro Super Thermo Powder. The original version with DMAA has not been manufactured or distributed since early 2013. Out of an abundance of caution, the company has ceased domestic distribution of OxyElite Pro with the Purple Top and OxyElite Pro Super Thermo Powder until the investigation has been completed. The company continues to believe these versions are safe and are not the cause of the cluster of liver toxicity that has occurred in Hawaii.”
However, the FDA reported on Monday that they sent a letter to the USPlabs located in Dallas, Tex., notifying them that if they did not initiate a voluntary recall of the product, they could “order the company to immediately stop distributing the dietary supplements and immediately notify other parties to stop distributing the dietary supplements.” Products involved in the recall include OxyElite Pro Super Thermo capsules, OyElite Pro Ultra-Intense Thermo capsules and OxyElite pro Super Thermo Powder.
If consumers currently are taking any of the aforementioned products, they are advised to discontinue use as the investigaton continues. Those who believe they may have been harmed from taking the dietary supplement should contact their health care provider and report illness they believe is related to the products to the FDA or 1-800-FDA-1088 or http://www.fda.gov/. Below is a full list of the products affected by the recall, according to the FDA:
OxyElite Pro Super Thermo capsules 2 count capsules
- UPC #094922417275 10 count capsules
- UPC #094922417251 10 count capsules
- UPC #094922417268 21 count capsules
- UPC #094922426604 90 count capsules
- UPC #094922395573 90 count capsules "Pink label"
- UPC #094922447906 180 count capsules
- UPC #094922447852
OxyElite Pro Ultra-Intense Thermo capsules 3 count capsules
- UPC #094922447883 3 count capsules
- UPC #094922447876 90 count capsules
- UPC #094922395627 180 count capsules
- UPC #094922447869
OxyElite Pro Super Thermo Powder Fruit Punch 0.15 oz
- UPC #094922417237 Fruit Punch 0.15 oz
- UPC #094922447517 Fruit Punch 4.6 oz
- UPC #094922426369 Fruit Punch 5 oz.
- UPC #094922447487 Blue Raspberry 4.6 oz
- UPC #094922426376 Grape Bubblegum 4.6 oz
- UPC #094922447500 Green Apple 4.6 oz.
- UPC #094922426499
© Copyright IBTimes 2024. All rights reserved.












