FDA: Dissolvable Tobacco Less Dangerous But Could Lead To More Users
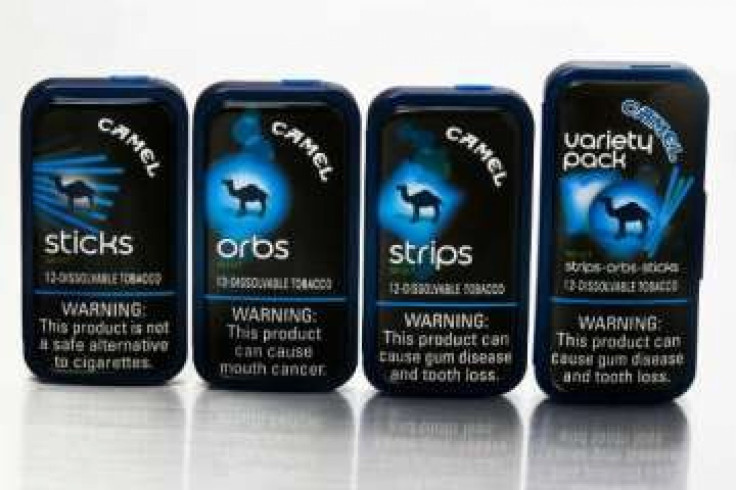
Tobacco that dissolves on the tongue may reduce health risks associated with smoking cigarettes, but could also increase the overall number of tobacco users, according to a Food and Drug Administration panel report.
Dissolvable tobacco is made from finely milled tobacco formed into tablets, strips or sticks that typically contain between 1 and 3 milligrams of nicotine and take 10-30 minutes to dissolve in the mouth. A typical cigarette contains 1-2 milligrams of nicotine.
Smokefree regulations and changing social norms have narrowed the range of venues where smoking is allowed and acceptable, committee members wrote in the report.
Based on understanding of the delivery of toxins to cigarette smokers, exclusive use of (dissolvable tobacco products) should be less hazardous than regular smoking of cigarettes now marketed in the United States, committee members wrote.
If smokers switch to dissolvable tobacco product, it could greatly reduce the risk of disease, according to the report.
At least one dissolvable tobacco makers responded favorably to the report.
At R.J. Reynolds Tobacco Company it is a guiding principle and belief that adult tobacco consumers have a right to be fully and accurately informed about the risks of serious diseases, the significant differences in the comparative risks of different tobacco and nicotine-based products, and the benefits of quitting, the company released in a statement. This information should be based on sound science. The report released by the Tobacco Product Scientific Advisory Committee (TPSAC) underscores the importance and relevance of this principle.
Despite fewer health effects, the American Cancer Society still considers dissolvable tobacco a danger.
We don't yet know how safe dissolvables are, the organization warned in a 2011 statement. Certainly, as with all smokeless tobacco products, they will be less lethal than smoked cigarettes, and smokers should certainly continue to be made aware of that. But we do know that other smokeless tobacco products are associated with increased risk of oral cancers, the potential for increased pancreatic cancer risk, and an increased risk of cardiovascular disease.
Surveys in Indiana and Virginia show that the widespread availability of dissolvable tobacco increased tobacco use as and created the potential to draw teens and adolescents to the product, according to the report.
Tobacco use is the leading preventable cause of death in the United States, according to the Centers for Disease Control and Prevention. Over 400,000 people are killed each year in the U.S. from tobacco use.
More information is needed before the FDA can create any regulation or recommendations about dissolvable tobacco, committee members noted. Long-term studies recommended included underage use, perception, health effects and effect on smoking and other tobacco product use. In addition, product contents and labeling need to be standardized across the industry, committee members argued
The FDA said they will continue to monitor all available scientific material and determine what future actions, if any, are needed to regulate the product.
© Copyright IBTimes 2024. All rights reserved.











