Morning After Pill Restrictions Upheld After Obama Administration Blocks FDA
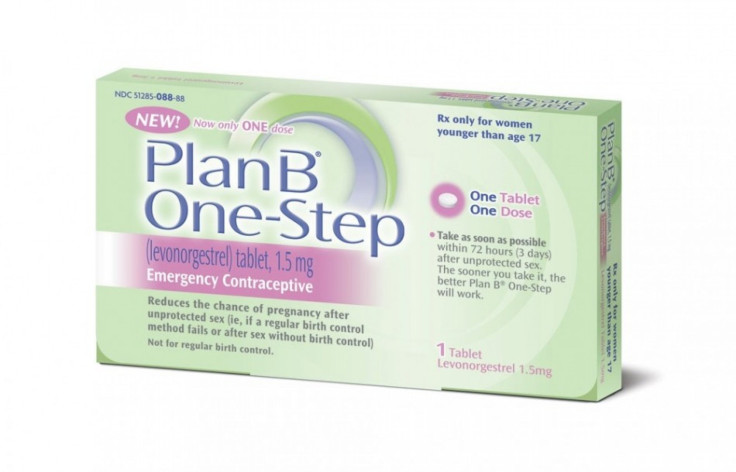
U.S. Health and Human Services Secretary Kathleen Sebelius intervened at the eleventh hour to stop the Food and Drug Administration (FDA) from lifting the age limit required to purchase Plan B One-Step, the nation's first over-the-counter emergency contraceptive, as well as allowing drugstores to move the birth control method onto shelves next to condoms.
The FDA was preparing to lift the age limit on Plan B, which is currently on available without a prescription to women over 17 who have a government-issued photo ID to confirm their age. Sebelius' decision was shocking to many who have participated in a nearly decade-long fight to allow the over-the-counter sale of medications that can prevent pregnancy if taken soon enough after participating in unprotected sex, something several women's health advocates have argued could cut down on the number the number of unplanned pregnancies in the U.S.
Although FDA Commissioner Margaret HamburFDA g had signaled she was planned to approve a request by Teva Pharmaceutical Industries, the maker of Plan B, to make it available for over-the-counter use of women of all ages, she acquiesced to Sebelius'-technically her boss - decision.
In a statement, Hamburg wrote that the Center for Drug Evaluation and Research determined that Plan B is safe and effective for adolescents and that they are capable of using the product safely, with an understanding that it is not meant for routine use.
I reviewed and thoughtfully considered the data, clinical information, and analysis provided by CDER, and I agree with the Center that there is adequate and reasonable, well-supported, and science-based evidence that Plan B One-Step is safe and effective and should be approved for nonprescription use for all females of child-bearing potential, Hamburg wrote, adding that Sebelius is able to invoke her authority on the issue under the Federal Food, Drug and Cosmetic Act.
Plan B One-Step will remain on the market and will remain available for all ages, but a prescription will continue to be required for females under the age of 17, Hamburg said.
In another statement, Sebelius wrote that Teva's application for Plan B's over-the-counter availability did not adequately demonstrate that there is enough evidence to show that those who use this medicine can understand the label and use the product appropriately.
Specifically, Sebelius said that because some girls as young as 11 are physically capable of bearing children, there is not enough evidence to suggest they would be capable of using Plan B as directed.
It is common knowledge that there are significant cognitive and behavioral differences between older adolescent girls and the youngest girls of reproductive age, Sebelius said.
Women's groups have spoken out against the Obama administration, arguing that Sebelius was directed to reject Plan B's wider availability for political gain as the 2012 presidential election approaches.
What else can this be but politics? Cynthia Pearson, the executive director of the National Women's Health Network, an advocacy group that supports making Plan B available to all ages, told The Associated Press. It's not science. It's not medicine. It's not women's health.
The FDA initially approved Plan B as a prescription-only medication in 1999. In 2003, the Women's Capital Corporation filed an application change the status of Plan B from prescription-only to available over-the-counter for all age groups, a proposal the agency has battled on an on-and-off basis for nearly a decade.
© Copyright IBTimes 2024. All rights reserved.











