2013 Nobel Prize In Physiology Or Medicine Awarded To Trio For Fundamental Research Into Cellular Transport
This year’s Nobel Prize in Medicine went to three researchers who mapped the mechanisms by which cells coordinate their transportation systems. The three scientists all came at the same issue from different angles – proving that there’s more than one road to discovery.
Just as a factory has to arrange for specific packages to be delivered to the right customers, the cell has to make sure that the molecules it produces arrive at the right destination. Over the past few decades, Randy Schekman of the University of California, Berkeley, James Rothman of Yale University, and Thomas Sudhof of Stanford University all unlocked different aspects of this mystery. On Monday, the Nobel Assembly at Sweden’s Karolinska Institute awarded them one of science’s highest honors (plus $1.2 million) for their work.
“Through their discoveries, Rothman, Schekman and Sudhof have revealed the exquisitely precise control system for the transport and delivery of cellular cargo,” the Nobel committee said in a press release. “Disturbances in this system have deleterious effects and contribute to conditions such as neurological diseases, diabetes and immunological disorders.”
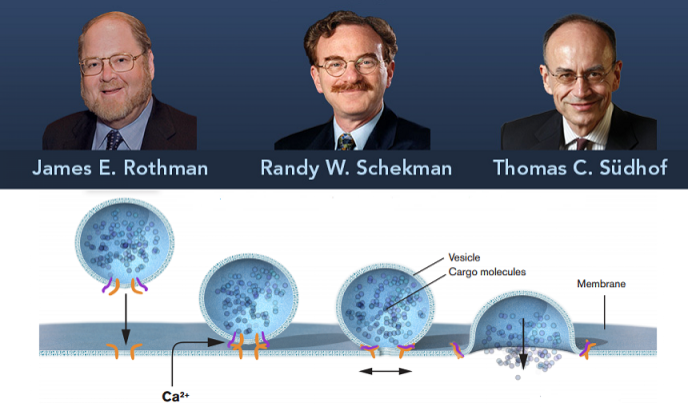
Much of intracellular transport relies on little bubble-like transport structures called vesicles. Vesicles bud from one cellular structure, cross the fluid that fills the cell, and fuse with the membrane of another structure or with the membrane that surrounds the cell like a skin.
"The discoveries of Schekman, Rothman and Sudhof elucidated the molecular mechanisms that during the formation of the [vesicles] ensure the selection of the correct cargo, target the vesicles to the appropriate receiving compartment, and control their fusion with its limiting membrane," New York University cell biologist David Sabatini wrote in an email.
In the 1970s, Schekman discovered the genetic marching orders that yeast cells use to control vesicles. One key paper came in 1979, in the Proceedings of the National Academy of Sciences. Schekman and colleague Peter Novick (now at the University of California San Diego) were looking at mutated yeast cells that could not properly secrete acids. They found that in these mutants, vesicles start piling up in the cell membrane – “a situation resembling a poorly planned public transport system,” according to the Karolinska Institute. By studying mutated cells, Schekman was able to zero in on the genes that were important in directing this cellular traffic.
Rothman first encountered the concept of cellular secretion in medical school at Harvard.
“What an astonishing process,” he wrote in an essay for Nature Medicine in 2002 [PDF]. “How could cells make vesicles from membranes? How could each vesicle know where to go? How could it fuse? It was particularly astonishing because at the time it was not even possible to begin to imagine the form a molecular solution might take.”
Rothman and his colleagues studied the Golgi apparatus, a structure inside the cell that serves as a kind of tiny post office: It processes vesicles from other organelles, packaging and “labeling” them so they can reach their final destination. In one key 1984 paper published in the journal Cell [PDF], Rothman and his team described how vesicles bind to specific spots on the Golgi apparatus, which led to later insights into how vesicles bind to a cell’s membrane to release their contents. Some of the genes their team found to be involved in mammalian cell transport directly corresponded to genes discovered by Schekman in yeast, hinting at a widely conserved method that endured across the biological kingdom.
Sudhof was specifically interested in vesicle trafficking as it relates to nerve cells. The neurotransmitters that drive signals in your brain are carried from nerve to nerve by vesicles, and their delivery is controlled by certain signals. Sudhof worked out how calcium ion signaling plays into the timing of vesicle transport in nerves. He and Rothman shared the Kavli Prize for neuroscience in 2010.
The trio’s research on cellular transportation laid the foundation for much of modern medicine and biological research.
"You can't understand anything about how the brain works without understanding this process,” Jeremy Berg, director of the University of Pittsburgh’s Institute for Personalized Medicine, told Reuters. “You can't understand anything about hormone secretion without understanding this process… there are probably literally thousands of laboratories around the world whose work would not be taking place the way it is without their work."
© Copyright IBTimes 2024. All rights reserved.





















