Bioluminescence In The City: How Glowing Eels And Green Jellyfish Could Light The Future
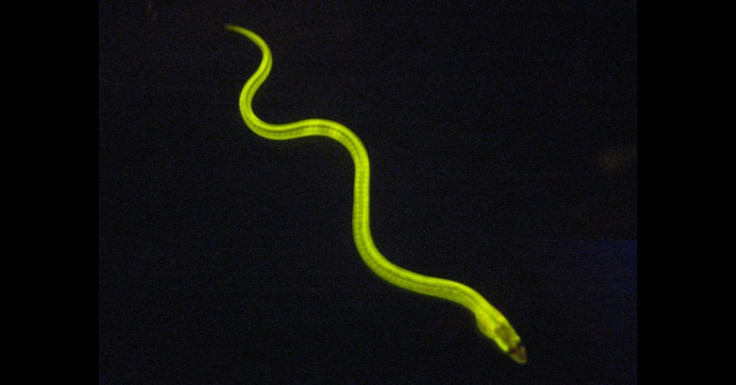
Japanese scientists recently relayed in the journal Cell that they’ve discovered a protein in the muscle fibers of the freshwater eel Anguilla japonica (which you may be more familiar with as a sushi ingredient) that glows greenish-yellow under blue light. The glowing eel is just one of many creatures that exhibit bioluminescence, the natural production of light.
Most current applications of fluorescent proteins are concerned with the very small, but some researchers are thinking bigger. Unlike a light bulb, this way of producing light creates very little wasted heat, and doesn't require vast amounts of fossil fuels to power. Could the way to the future be illuminated by glowing eels or flashing fireflies?
The Japanese team dubbed their recently discovered eel protein UnaG. Other glowing proteins have been isolated from other animals – most notably green fluorescent protein or GFP, taken from the crystal jellyfish Aequorea victoria. GFP is already an essential element of many a researcher’s tool kit, used to “tag” other proteins within living cells to track the expression of genes. But this is the first time that a fluorescent protein has been found in a vertebrate creature.
Eels and jellyfish aren’t the only creatures that make their own light. As summer blooms, some people may get a glimpse of a firefly’s glow. The firefly, probably the most familiar examples of natural light production, or bioluminescence, makes its light with a carefully controlled chemical reaction. The key ingredient is an enzyme called luciferase, which catalyzes a reaction that leads to producing light. Fireflies can control the reaction through a process involving the restriction of oxygen, but it's still not completely known how they accomplish this.
Luciferase can be used, like GFP, to report on the activity of gene expression inside cells. Firefly bioluminescence may have also contributed to a very early kind of photography. One researcher has claimed that the Italian artist Caravaggio could have used crushed fireflies to treat his canvases to make them light-sensitive, “fixing” an image that then allowed him to rough in an image without making an underlying sketch.
And if you can’t make your own light, why not capture some, as the bobtail squid does? This animal cultivates a crop of bioluminescent bacteria (Vibrio fischeri) inside of a special organ its mantle. The bobtail squid’s light organ is chiefly used to hide it from predators; directed downward, it hides the silhouette of the squid from creatures below it.
Right now, one of the bigger limitations to using bioluminesence for actual light is that fluorescent proteins simply aren't strong enough to provide a meaningful light source. Biological engineering, however, could pump up the wattage. In 2010, undergraduate students from the University of Cambridge modified genes from V. fischeri and fireflies to boost their shining power and inserted them into an E. coli bacteria. A colony of these bacteria large enough to fill a wine bottle produced enough light to read by. The team also envisions introducing these souped-up light-producing proteins into trees, which could then become natural streetlights.
"That was exciting for us, but it is not going to replace road signs anytime soon," student Theo Sanderson told Popular Science.
Other potential applications of bioluminesence include creating plants that glow different colors to signal farmers about dehydration, or disease, or pests. A potato that lights up to let a farmer know that it's thirsty could help prevent water waste.
Bio-light probably won't be threatening the supremacy of the light bulb any time soon. But the more that we learn about bioluminescence, the more one starts to think about other possibilities that living neon light affords. UC San Diego researchers have already created a billboard made of millions of glowing bacteria – now imagine seeing Times Square lit up by colonies of microorganisms, no electricity required!
© Copyright IBTimes 2024. All rights reserved.





















