A Vaccine For Malaria? Some Scientists Are Closer Than Ever Following Groundbreaking Clinical Trials
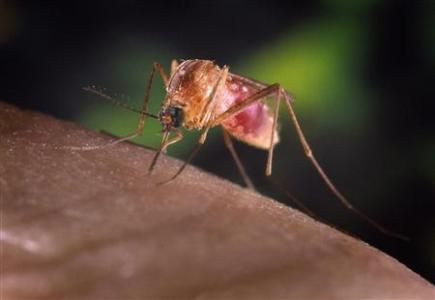
Scientists have made an unprecedented breakthrough in the fight against malaria this week when early trials of a new vaccine yielded stellar results. The study was tiny and the research is far from over. But, if the vaccine -- known as PfSPZ -- lives up to its potential, one of the world's most deadly diseases may finally meet its match.
The Phase I clinical trial tested 57 people, 40 of whom received varying doses of the vaccine. All participants were then infected by the malarial parasite, and most of those who had received no or low doses of the vaccines came down with the disease. Of the 15 people who received relatively high doses, only 12 contracted malaria. And, most encouragingly, of the six volunteers who received the highest dosage -- five shots of PfSPZ -- not a single one was infected with malaria.
"While we’re still in the early stages of testing, we believe this vaccine will be used to eliminate malaria,” said Stephen L. Hoffman, CEO of Rockville, Md.-based Sanaria, which developed and tested the PfSPZ vaccine, in a statement.
The vaccine will have to jump through many more hoops yet before it can be brought to market. Future trials -- planned to take place in Africa, the U.S. and Europe -- will have to be much larger, and they must be held over a longer period of time in order to show that the vaccine can remain effective long after it is administered.
PfSPZ also faces logistical challenges. It must be delivered into the bloodstream, making it more difficult to administer than a simple subcutaneous shot, which presents a potential hurdle in places with substandard health care practices and facilities.
Nevertheless, experts at Sanaria are enthusiastic about their groundbreaking results.
“It’s reasonable to suggest that within three-to-five years, a safe, reliable vaccine could be a commercial reality and provide medical benefit to a huge population,” Hoffman said.
Malaria in humans is spread by mosquitoes carrying the Plasmodium parasite; it is endemic in 106 countries. More than 90 percent of malaria deaths occur in sub-Saharan Africa, but the disease is also a scourge in some parts of Asia and Latin America.
The most recent data from the World Health Organization, or WHO, shows that in 2010, about 219 million people were infected by malaria. An estimated 660,000 -- mostly young children -- died after contracting the disease. Symptoms often include fever, chills, vomiting and diarrhea while severe cases can lead to delirium, organ failure and seizures.
Current treatments to fight malaria consist mostly of oral medications, some of which are preventative while others are meant to ease symptoms.
The most popular treatments for malaria come from artemisinin, a plant-based compound that has helped to cut down malaria infections worldwide. According to WHO, global malaria mortality rates have decreased by more than 25 percent since 2000.
But artemisinin-based drugs only work if they're of a high quality while substandard versions can actually build up the malaria parasite's resistance, strengthening the deadly disease over time. This May, a Lancet study found that more than one-third of the drugs distributed to fight malaria in Southeast Asia and sub-Saharan Africa over the past decade years were either fake or of poor quality.
A vaccine would help to circumvent this very serious problem, but only if it can prove its viability over numerous clinical trials. Sanaria hopes it will be the company to succeed where others have failed.
“Scientists have struggled to produce an effective malaria vaccine for more than three decades,” Hoffman said, in the statement. “These results show that we have a safe, successful, injectable vaccine that has the potential to save millions of lives.”
© Copyright IBTimes 2025. All rights reserved.





















