What Is Metallic Hydrogen? Doubts Raised Over Reported Creation Of Extremely Rare And Potentially Valuable Material
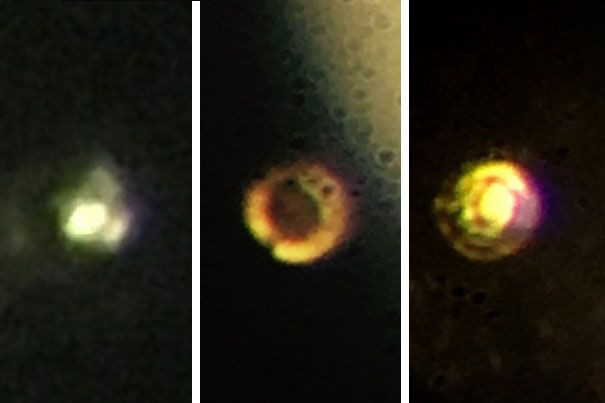
Ever since the existence of metallic hydrogen was theorized over 80 years ago, the material has been the Holy Grail of condensed-matter physics. Now, two researchers from Harvard University say, in a study published Thursday in the journal Science, that they have succeeded in creating this extremely exotic substance — although many physicists remain skeptical of the claims.
“It's the first-ever sample of metallic hydrogen on Earth, so when you're looking at it, you're looking at something that's never existed before,” study co-author Isaac Silvera, a professor of the natural sciences at Harvard, said in a statement.
So what exactly is metallic hydrogen, and why is this breakthrough important?
Under normal conditions, hydrogen — the lightest element — is a colorless gas. However, in 1935, physicists Eugene Wigner and Hillard Huntington predicted that if subjected to sufficiently high pressure — say 250,000 times Earth’s atmospheric pressure — hydrogen would turn into a solid metal.
“One prediction that's very important is metallic hydrogen is predicted to be meta-stable,” Silvera said. “That means if you take the pressure off, it will stay metallic, similar to the way diamonds form from graphite under intense heat and pressure, but remains a diamond when that pressure and heat is removed.”
In their experiment, Silvera and his colleague Ranga Dias used a device known as a diamond anvil cell and subjected a tiny hydrogen sample to a pressure of 495 gigapascals — greater than the pressure at the centre of the Earth. As the researchers explained in the study, at these pressures, solid molecular hydrogen breaks down, and the tightly bound molecules transform into atomic hydrogen, which is a metal.
The creation of metallic hydrogen could, in addition to furthering our understanding of the general properties of hydrogen, pave the way for the creation of potentially revolutionary new materials, including something that has been long sought after — room temperature superconductors.
Superconductors — materials that can conduct electricity with zero resistance and thereby zero loss of energy — have a wide range of uses, the most well-known being their application in Magnetic Resonance Imaging (MRI) machines. However, the potential of superconducting materials is limited by the fact that they need to be cooled down to nearly -270 degrees Celsius (-454 degrees Fahrenheit) in order to function — something that requires a lot of energy and is therefore extremely expensive.
If, as researchers predicted in the past, metallic hydrogen can act as a superconductor at room temperature, it could one day be used to create superconducting electrical wires, and to drastically improve the efficiency of many electronic devices currently in use.
In addition, metallic hydrogen can also be used as an extremely powerful rocket propellant.
“It takes a tremendous amount of energy to make metallic hydrogen,” Silvera said. “And if you convert it back to molecular hydrogen, all that energy is released, so it would make it the most powerful rocket propellant known to man, and could revolutionize rocketry.”
The reactions from other physicists, however, ranged from mildly skeptical to vociferously critical. Many questioned how the Harvard researchers managed to generate the immense pressure detailed in the study on their first try, and whether the experiment was reproducible.
“I don’t think the paper is convincing at all,” Paul Loubeyre, a physicist at France’s Atomic Energy Commission told Nature News. “If they want to be convincing, they have to redo the measurement, really measuring the evolution of pressure.”
© Copyright IBTimes 2025. All rights reserved.






















