2016 Nobel Prize In Chemistry Awarded To Trio Of Scientists Who Created World's Smallest Machine

Jean-Pierre Sauvage, Sir Fraser Stoddart and Bernard Feringa were awarded the 2016 Nobel Prize for Chemistry at a conference in Stockholm, Sweden for creating the world’s smallest machines. The trio of European laureates, who each worked on a different component of the project, will split the 8 million Swedish kronor prize equally.
The tiny machines are built on a molecular scale and are a thousand times thinner than a strand of hair, reports the BBC. According to the Nobel Committee, the scientists “have mastered motion control at the molecular scale.”
“The laureates have opened this entire field of molecular machinery,” said Göran Hansson, secretary general of the Swedish Academy of Sciences, reports The Guardian. “They have shown it is possible to make a machine at the molecular scale.”
It all started in 1983 when Sauvage, now a professor emeritus at the University of Strasbourg in France, created a molecular chain—or a catenane—by linking two rings using a copper ion.
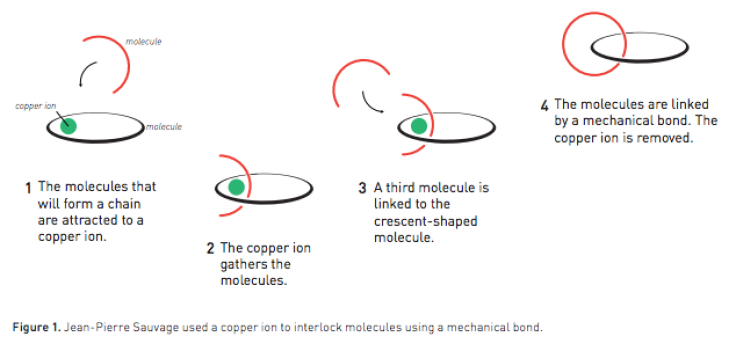
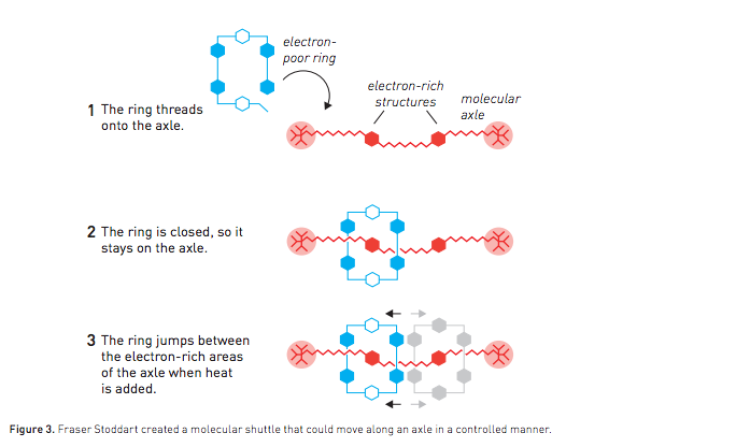
In 1991, Stoddart, who is affiliated with Northwestern University, realized he could use an electron-poor open ring and electron-rich rod to create a rotaxane or a ring on an axle.
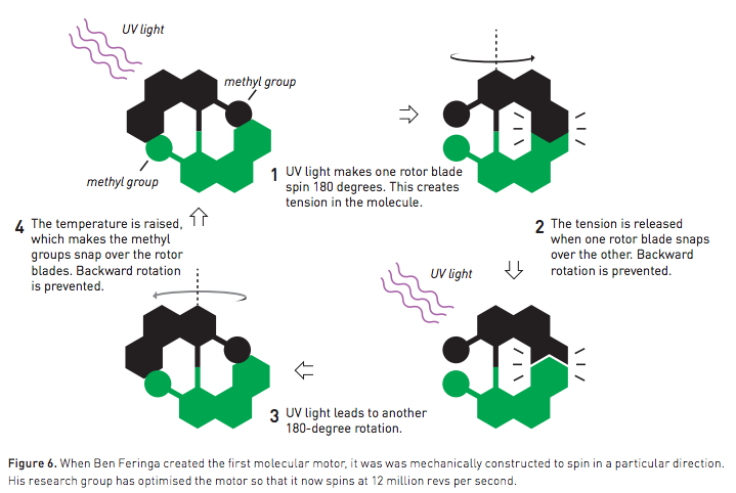
Feriga, a professor at the University of Groningen in the Netherlands, designed the first molecular motor in 1999 that makes a motor blade spin in one direction using pulses of light.
“I feel a little bit like the Wright brothers who were flying 100 years ago for the first time and people were saying why do we need a flying machine?” Feringa told the Nobel committee in a telephone interview. “And now we have the Boeing 747 and the Airbus. This is a bit how I feel. The opportunities are great because if you think what kinds of materials we can make these days with the chemistry and when you are able to introduce dynamic function, there is endless opportunity. We will build smart materials in the future, that is a big opportunity, materials that will adapt themselves, that will have properties that can change because they pick up a signal. You can think of nanoscale energy converters, tiny machines that can store energy and can use that energy. It opens up a whole new world of nanomachines.”
The application of this technology is vast ranging from drug delivery and smart materials to artificial life. Currently, the technology is being utilized to create medical micro-robots and self-healing materials.
“If you want infrastructure that looks after itself - and I think we do - I’m pretty sure we’re going to be moving towards self-healing systems,” said Mark Miodownik, professor of materials and society at the University College London. “We’ll have plastic pipes that can repair themselves or a bridge that when it gets cracked has these machines that rebuild the bridge at a microscopic scale. It’s just beginning. The potential is really immense.”
The Nobel Prize website states:
"2016's Nobel Laureates in Chemistry have taken molecular systems out of equilibrium's stalemate and into energy-filled states in which their movements can be controlled. In terms of development, the molecular motor is at the same stage as the electric motor was in the 1830s, when scientists displayed various spinning cranks and wheels, unaware that they would lead to electric trains, washing machines, fans and food processors. Molecular machines will most likely be used in the development of things such as new materials, sensors and energy storage systems."
© Copyright IBTimes 2024. All rights reserved.












