MIT Researchers Discover New Drug that May Cure Nearly All Viral Infection
Researchers at the Massachusetts Institute of Technology, or MIT, have developed what could be a groundbreaking drug that seems effective at curing nearly any viral infection to include the common cold, influenza and other such illnesses.
The drug, dubbed DRACO, or Double-stranded RNA Activated Caspase Oligomerizers, works by targeting a type of RNA produced only in virus-infected cells. Because it's so broad-spectrum, DRACO could potentially be used to fight outbreaks of new viruses, such as the 2003 severe acute respiratory syndrome, or SARS, outbreak, says Todd Rider, who invented the drug. Rider is a senior staff scientist in MIT's Lincoln Laboratory's Chemical, Biological, and Nanoscale Technologies Group.
"In theory, it should work against all viruses," Rider says in a statement posted on MIT's Web site.
A paper on the drug can be read in the journal PLoS One.
According to a press release on DRACO, Rider had the idea to try and develop a broad-spectrum antiviral therapy some 11 years ago. This was after inventing CANARY (Cellular Analysis and Notification of Antigen Risks and Yields), a biosensor that can rapidly identify pathogens.
"If you detect a pathogenic bacterium in the environment, there is probably an antibiotic that could be used to treat someone exposed to that, but I realized there are very few treatments out there for viruses," Rider says.
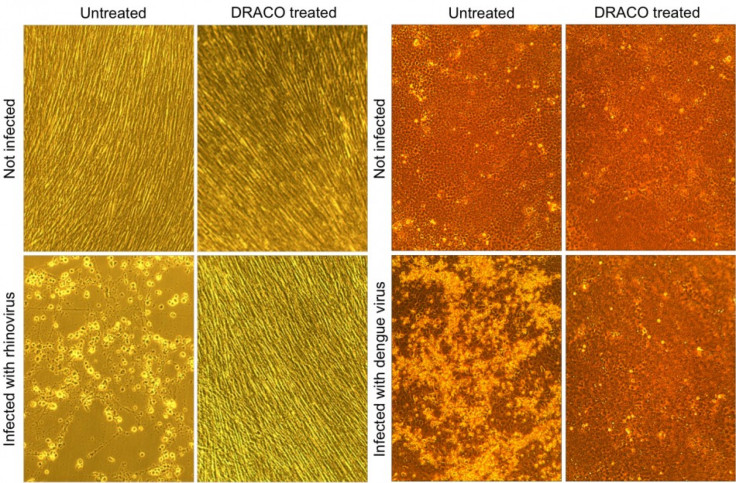
He drew inspiration for DRACO from living cells' own defense systems. Researchers tested the drug against 15 viruses and found it was effective against all of them to include rhinoviruses that cause the common cold, H1N1 influenza, a stomach virus, a polio virus, dengue fever and several other types of hemorrhagic fever.
When viruses infect a cell, they take over its cellular machinery to use for their own purpose. Their purpose is to create more copies of the themselves, and during this process, the viruses make long strings of double-stranded RNA (dsRNA), which isn't found in human or other animal cells, a press release notes.
The human cells, as part of their natural defenses against viral infection, have proteins that latch onto dsRNA, which then sets off a cascade of reactions that prevents the virus from replicating itself. But many viruses can outsmart that system by blocking one of the steps further down that cascade.
So Rider had the idea to combine a dsRNA-binding protein with another protein that induces cells to undergo apoptosis (programmed cell suicide) — launched, for example, when a cell determines it's en route to becoming cancerous. And so, when one end of the DRACO joins to dsRNA, it signals the other end of the DRACO to initiate cell suicide, the press release states.
Karla Kirkegaard, professor of microbiology and immunology at Stanford University, says combining those two elements is a "great idea" and a very novel approach.
"Viruses are pretty good at developing resistance to things we try against them, but in this case, it's hard to think of a simple pathway to drug resistance," she says in a statement.
Researchers say most of the tests reported in this study were done in human and animal cells cultured in the lab, but the they also tested DRACO in mice infected with the H1N1 influenza virus. When mice were treated with DRACO, they were completely cured of the infection. The tests also showed that DRACO itself is not toxic to mice.
Researchers are now testing DRACO against more viruses in mice and beginning to get promising results.
Rider says he hopes to license the technology for trials in larger animals and for eventual human clinical trials.
© Copyright IBTimes 2024. All rights reserved.






















