Self-Healing Battery: Scientists Invent Next-Generation Battery For Future Devices
Could this be the invention that will allow electric vehicles to permanently displace gasoline-powered cars?
A group of scientists from Stanford University and the Department of Energy's SLAC National Accelerator Laboratory has developed a battery electrode that can heal itself, potentially offering a commercially viable path for making the next generation of lithium-ion batteries to be used in electric cars, cell phones and other devices.
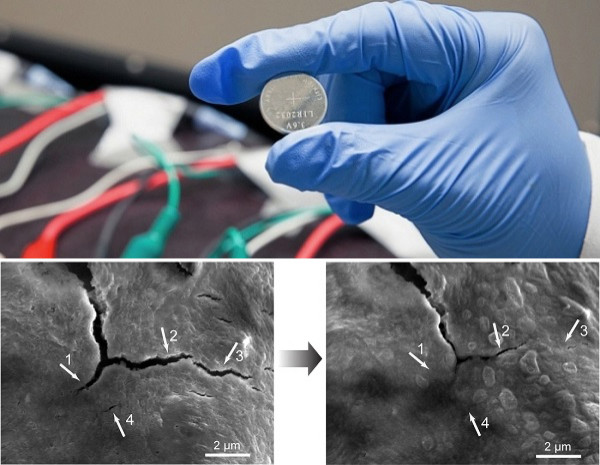
According to scientists, the secret behind the newly invented battery is a stretchy polymer that coats the electrode, binds it together and instinctively heals small cracks that develop while the battery is in use.
"Self-healing is very important for the survival and long lifetimes of animals and plants," Chao Wang, a postdoctoral researcher at Stanford and one of two principal authors of the paper, said in a statement. "We want to incorporate this feature into lithium-ion batteries so they will have a long lifetime as well."
For the project, Chao added tiny nanoparticles of carbon to the polymer so it would conduct electricity.
"We found that silicon electrodes lasted 10 times longer when coated with the self-healing polymer, which repaired any cracks within just a few hours," Zhenan Bao, a Stanford professor, said in a statement.
The new battery electrodes currently work for about 100 charge-discharge cycles without significantly losing their energy-storage capacity. However, scientists are aiming to enhance the capability to about 500 cycles for cell phones and up to 3,000 cycles for an electric vehicle.
According to scientists, one of the most promising electrode materials is silicon, which can soak up lithium ions from the battery fluid during charging, and then release them when the battery is put to work. However, silicon electrodes expand to three times their normal size and shrink back down again each time the battery charges and discharges, leading the material to crack soon and diminish the battery's performance.
“To make the self-healing coating, scientists deliberately weakened some of the chemical bonds within polymers – long, chain-like molecules with many identical units,” a Stanford University statement read. “The resulting material breaks easily, but the broken ends are chemically drawn to each other and quickly link up again, mimicking the process that allows biological molecules such as DNA to assemble, rearrange and break down.”
A research paper related to the project was published online on Nature Chemistry on Sunday.
© Copyright IBTimes 2024. All rights reserved.






















