World's Only Sample Of Metallic Hydrogen — Holy Grail Of Condensed-Matter Physics — Has ‘Disappeared’
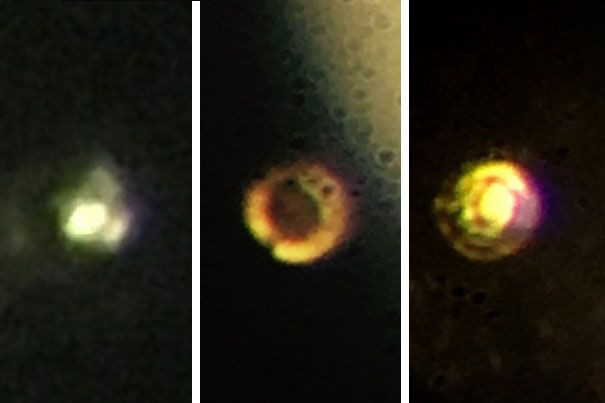
Last month, two researchers from Harvard University created waves in the field of condensed-matter physics when they claimed they had succeeded in creating an extremely exotic substance called metallic hydrogen. Their achievement, the researchers said, confirmed that this substance — the metallic form of an element that, under normal conditions, is a gas — did really exist outside of theory books.
The sample of metallic hydrogen these researchers created was being stored at temperatures close to absolute zero in a diamond vice. But it has now been revealed that the sample has either degraded or been misplaced.
“Basically, it's disappeared,” Isaac Silvera, a professor of the natural sciences at Harvard, who was one of the scientists involved in the creation of the sample, told ScienceAlert in a report published Wednesday. “It's either someplace at room pressure, very small, or it just turned back into a gas. We don't know.”
In their experiment, detailed in a study published in the journal Science, Silvera and his colleague Ranga Dias used a device known as a diamond anvil cell and subjected a tiny hydrogen sample to a pressure of 495 gigapascals — greater than the pressure at the centre of the Earth. At these pressures, solid molecular hydrogen breaks down, and the tightly bound molecules transform into atomic hydrogen, which is a metal.
However, when the researchers used a low-powered laser to measure the pressure of the system once more earlier this month, the energy from the laser reportedly destroyed the diamond vice.
“I’ve never seen a diamond shatter like that. It was so powdered on the surface, it looked like baking soda or something like that,” Silvera told the Independent.
The news assumes special significance in the light of the fact that Silvera’s and Dias’ claims had elicited scepticism and criticism from many corners. After the study was published, several physicists questioned how the researchers had managed, on their first try, to generate the immense pressure needed to create metallic hydrogen, and whether the experiment was reproducible.
“I don’t think the paper is convincing at all,” Paul Loubeyre, a physicist at France’s Atomic Energy Commission told Nature News. “If they want to be convincing, they have to redo the measurement, really measuring the evolution of pressure.”
For now, though, these questions will remain unanswered.
“We're preparing a new experiment to see if we can reproduce the pressures we achieved the first time, and reproduce our metallic hydrogen,” Silvera told ScienceAlert. “This disappearance doesn't say anything about the validity of the sample. Anyone who does high pressure works knows that you have failures like this.”
If the creation of metallic hydrogen — the “holy grail” of condensed-matter physics — is confirmed, it would not only significantly advance our understanding of the general properties of the element, it would also pave the way for the creation of revolutionary new materials, including room temperature superconductors. In addition, the material could be used as an ultralight and extremely powerful rocket fuel.
© Copyright IBTimes 2024. All rights reserved.






















