Scientists Develop Shape-Shifting Microbots That May Soon Be Used To Carry Out Precise Medical Operations
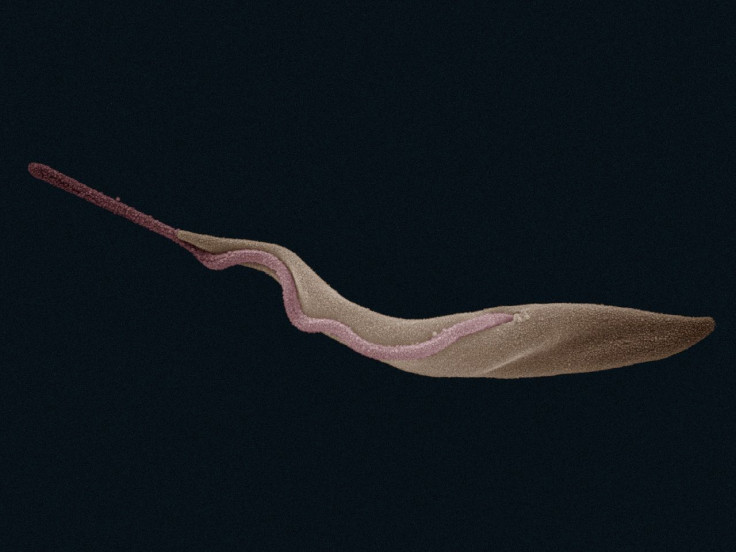
In a village in western Africa, a tsetse fly bites a human, injecting a swarm of the deadly sleeping sickness-causing parasite Trypanosoma brucei into the bloodstream. Once inside its human host, the parasite spreads through the body, rapidly using its flexible flagellum to propel itself, eventually hiding the structure inside its body to evade the host's immune system once it is safely ensconced.
Imagine a swarm of microscopic bots that mimics Trypanosoma brucei's behavior. In this case, though, instead of killing their hosts, the bots are designed to circulate through the bloodstream to perform highly-targeted drug deliveries and carry out invasive and delicate surgeries that may otherwise be too risky to perform.
This is what a team of scientists at the Swiss Federal Institute of Technology in Zurich (ETHZ) and the Swiss Federal Institute of Technology in Lausanne (EPFL) are working toward. And, in a new study published in the journal Nature Communications, these researchers have now described a new method for building such "microbots."
"We show that both a bacterium’s body and its flagellum play an important role in its movement,” EPFL scientist Mahmut Selman Sakar — a co-author of the study — said in a statement. "Our new production method lets us test an array of shapes and combinations to obtain the best motion capability for a given task. Our research also provides valuable insight into how bacteria move inside the human body and adapt to changes in their microenvironment."
According to the researchers, these microrobots — made of biocompatible hydrogel and magnetic nanoparticles — are soft, flexible, and motorless. The magnetic nanoparticles serve two purposes — they give the bots their shape and they make them move under the influence of an electromagnetic field.
"Building one of these microrobots involves several steps. First, the nanoparticles are placed inside layers of a biocompatible hydrogel. Then an electromagnetic field is applied to orientate the nanoparticles at different parts of the robot, followed by a polymerization step to 'solidify' the hydrogel. After this, the robot is placed in water where it folds in specific ways depending on the orientation of the nanoparticles inside the gel, to form the final overall 3D architecture of the microrobot," the researchers explained in the statement.
The prototype microbot, which the scientists hope would eventually be capable of performing precise operations like clearing up clogged arteries, also has a Trypanosoma-like flagellum that enables it to swim.
For now, though, the microbots are still in its early stages of development. The researchers are currently working to ensure that the bots, once inside the human body, do not cause any side-effects.
© Copyright IBTimes 2025. All rights reserved.




















